|
10.01.2018 00:57:00
|
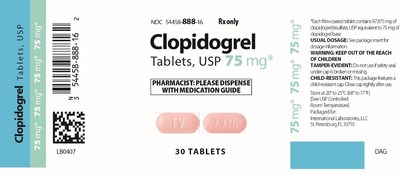
International Laboratories, LLC Issues Voluntary Nationwide Recall of one (1) Lot of Clopidogrel Tablets USP, 75 mg packaged in bottles of 30 tablets Due to Mislabeling
SEMINOLE, Fla., Jan. 9, 2018/PRNewswire/ -- International Laboratories, LLC is voluntarily recalling Lot# 117099A of Clopidogrel Tablets, USP 75 mg, packaged in bottles of 30 tablets, to the consumer level due to mislabeling. The product is labeled as Clopidogrel tablets USP 75 mg but may contain Clopidogrel 75 mg or Simvastatin Tablets USP 10 mg.
Missed doses of Clopidogrel increases the risk of heart attack and stroke which can be life threatening. Patients should not stop taking clopidogrel without talking to their prescribing physician. Additionally, unintentional consumption of simvastatin could include the common side effects associated with its use and may cause fetal harm when administered to a pregnant woman. Simvastatin occasionally causes myopathy which is a disease of the muscles. Finally, allergic reactions are also possible and could also be life threatening. International Laboratories, LLC also reports that to date, no complaints have been received related to this event detailing medical illnesses or harmful effects.
Clopidogrel Tablets USP 75 mgare a platelet inhibitor (blood thinner) indicated for the use in patient with acute coronary syndrome, recent MI, recent stroke, or established peripheral arterial disease. Clopidogrel tablets have been shown to reduce the rate of MI and stroke.
Clopidogrel 75mg Simvastatin 10 mg Tablet Description: Tablet Description light-pink to pink Light-pink, round capsule shaped "7153" on one side "TV" on one side "TV" on the other side "7314" on the other side
The product was distributed nationwide and delivered to the distribution centers inArkansas, Georgia, Indiana, California and Maryland, and distributed to retail stores in all US States.
International Laboratories, LLC is notifying distributors and customers by letter and is arranging for return of all recalled products. Consumers who have purchased this product should stop using and return the product to the location of purchase for a full refund. For questions regarding return of product please call Inmar at 855-258-7280 or via email internationallabs@inmar.com or by using mailing address Recall Coordinator 635 Vine St. Winston Salem, NC 27101 Inmar's business hours are (Monday – Friday 9 AM – 5 PM EST).
Consumers should also contact their physician or healthcare provider if they are experiencing any health concerns that may be related to taking or using this drug product.
Adverse reactions or quality problems experienced with the use of this product may be reported to the FDA's MedWatch Adverse Event Reporting program either online, by regular mail or by fax.
- Complete and submit the report Online: www.fda.gov/medwatch/report.htm
- Regular Mail or Fax: Download form www.fda.gov/MedWatch/getforms.htm or call 1-800-332-1088 to request a reporting form, then complete and return to the address on the pre-addressed form, or submit by fax to 1-800-FDA-0178
This recall is being conducted with the knowledge of the U.S. Food and Drug Administration.
![]() View original content with multimedia:http://www.prnewswire.com/news-releases/international-laboratories-llc-issues-voluntary-nationwide-recall-of-one-1-lot-of-clopidogrel-tablets-usp-75-mg-packaged-in-bottles-of-30-tablets-due-to-mislabeling-300580450.html
View original content with multimedia:http://www.prnewswire.com/news-releases/international-laboratories-llc-issues-voluntary-nationwide-recall-of-one-1-lot-of-clopidogrel-tablets-usp-75-mg-packaged-in-bottles-of-30-tablets-due-to-mislabeling-300580450.html
SOURCE International Laboratories, LLC
 Der finanzen.at Ratgeber für Aktien!
Der finanzen.at Ratgeber für Aktien!
Wenn Sie mehr über das Thema Aktien erfahren wollen, finden Sie in unserem Ratgeber viele interessante Artikel dazu!
Jetzt informieren!
Nachrichten zu Pentair PLCmehr Nachrichten
|
10:03 |
S&P 500-Papier Pentair-Aktie: So viel Gewinn hätte eine Pentair-Investition von vor einem Jahr eingebracht (finanzen.at) | |
|
19.11.24 |
S&P 500-Wert Pentair-Aktie: So viel Gewinn hätte ein Investment in Pentair von vor 10 Jahren abgeworfen (finanzen.at) | |
|
13.11.24 |
Freundlicher Handel in New York: S&P 500 mittags mit Gewinnen (finanzen.at) | |
|
12.11.24 |
S&P 500-Wert Pentair-Aktie: So viel Gewinn hätte eine Pentair-Investition von vor 5 Jahren eingebracht (finanzen.at) | |
|
05.11.24 |
S&P 500-Titel Pentair-Aktie: So viel Gewinn hätte eine Investition in Pentair von vor 3 Jahren abgeworfen (finanzen.at) | |
|
29.10.24 |
S&P 500-Wert Pentair-Aktie: So viel Gewinn hätte eine Investition in Pentair von vor einem Jahr abgeworfen (finanzen.at) | |
|
22.10.24 |
S&P 500-Wert Pentair-Aktie: So viel hätten Anleger mit einem Investment in Pentair von vor 10 Jahren verdient (finanzen.at) | |
|
21.10.24 |
Ausblick: Pentair gibt Ergebnis zum abgelaufenen Quartal bekannt (finanzen.net) |